Nurochek™
FDA-cleared portable EEG system for objective concussion assessment in approximately 2 minutes. No baseline required.
Product Overview
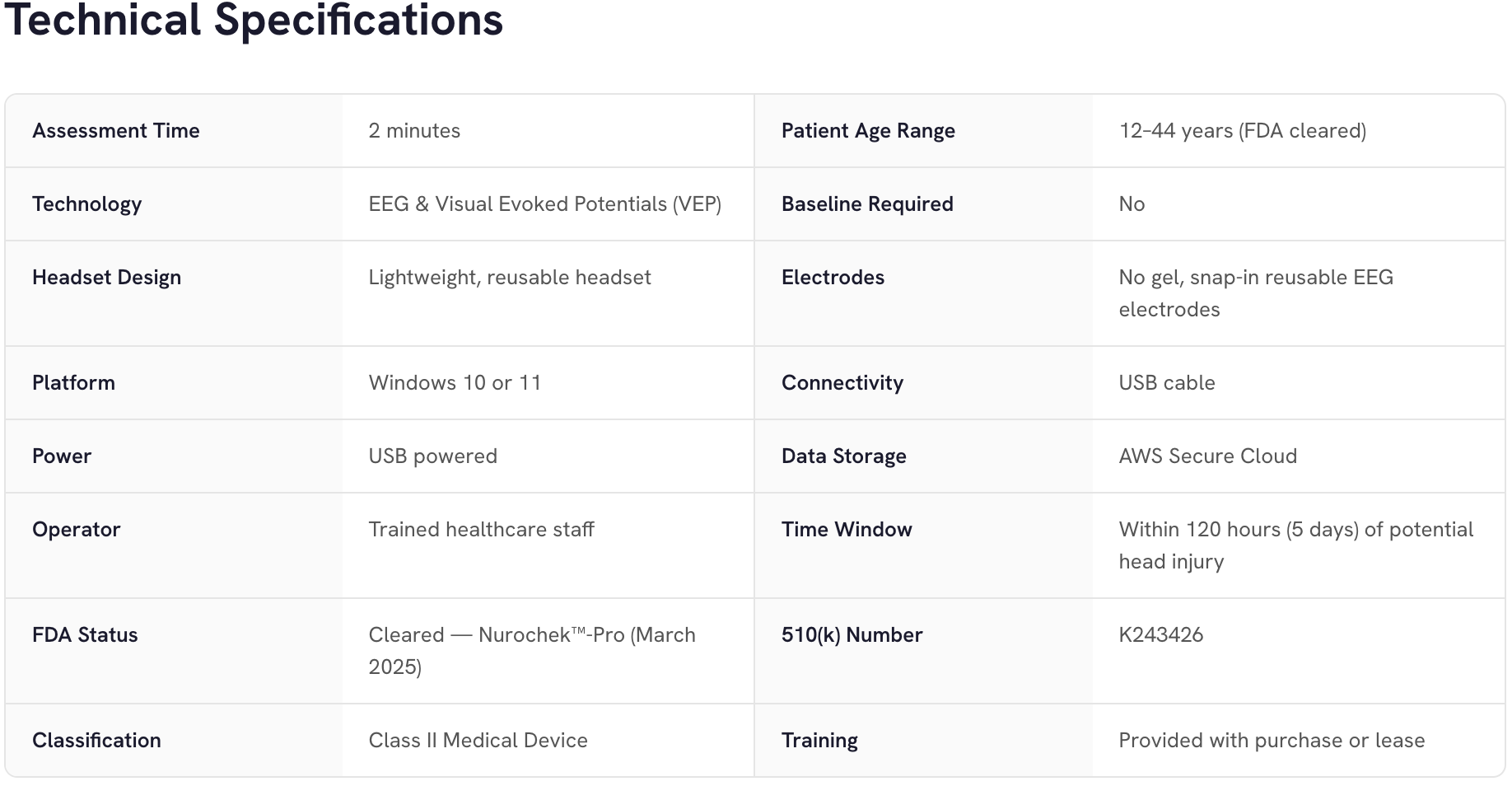
Nurochek™ is a portable, FDA-cleared brain assessment device used as an aid to diagnosis concussions that delivers objective brain function data in approximately two minutes, without requiring a baseline test.
Using EEG and Visual Evoked Potential (VEP) technology, Nurochek™ measures actual brain function—data that cannot be faked, minimized, or misreported. Proprietary algorithms analyze over 400,000 data points to produce an objective measure of brain function, supporting confident clinical decisions.
Designed for use in far-forward operations, athletic venues/sporting events, emergency departments, urgent care, rehab/physical therapy facilities, and other clinical environments, Nurochek™ is intended for use in healthcare facilities by or under the supervision of qualified healthcare professionals.




About Headsafe
Headsafe is a MedTech company that was founded in Sydney, Australia, with its U.S. headquarters in Lake Mary, Florida. Headsafe's growing U.S. presence is continuing its mission is to make concussion assessment faster, safer, and more consistent across points of care.
With U.S. and Global Operations led by CEO Craig Corrance, Headsafe is focused on supporting real-world adoption through practical implementation, training, and clinical workflow integration.
Important Use Information
Nurochek™ is FDA-cleared as an aid in the diagnosis of mild traumatic brain injury (mTBI, concussion) when used in conjunction with a standard neurological assessment. It is cleared for patients ages 12 to 44 years old, within 120 hours (5 days) of a potential head injury, and is intended for prescription use in healthcare facilities or by healthcare professionals.
Nurochek™-Pro is intended to be used as part of a comprehensive clinical evaluation and is not a standalone diagnostic device. Results should be interpreted by qualified healthcare providers in conjunction with clinical examination, patient history, and other diagnostic information.
Contraindications: Do not use if patient has a history of epilepsy and/or seizures, existing structural brain injury and/or condition, is legally blind, or has open head wounds/breached skin in contacting areas.
All necessary regulatory documentation, instructions for use, published studies, patient education tools, etc. (as applicable) can be provided upon request by contacting cs@spartanmedical.com or (888) 240-8091. We are available at any time to meet with providers and staff to answer any questions about Spartan Medical’s entire portfolio of advanced medical solutions.
Looking for more Software solutions?
Click on the below link(s) to see our entire portfolio



